Signaling, Contraction &
Response — Measured in Human Models.
NeuroMuscle™ is a fully human neuromuscular junction (NMJ)-on-a-chip platform that combines iPSC-derived motor neurons and 3D skeletal muscle within a microfluidic system to recreate functional neuromuscular signaling in vitro. Designed as a plug-and-play, plate-based system, NeuroMuscle™ operates like a standard multi-well culture plate—requiring no pumps, tubing, controllers, or specialized hardware—and integrates seamlessly into existing laboratory workflows.
Each disposable plate contains 20 independent, functional NMJs, enabling parallel testing across multiple conditions while supporting high-content imaging, functional assays, and automated analysis. The platform delivers quantitative, real-time readouts of neuromuscular communication and muscle contraction, providing insights that are difficult or impossible to obtain from traditional in vitro or animal models.
NeuroMuscle™ has been successfully transferred and established in external laboratories, including regulatory and industry environments, demonstrating robustness, reproducibility, and ease of implementation. The platform supports both in-house use and CRO-led study execution, with options for ready-to-use, pre-assembled NMJs.
Key Capabilities
1
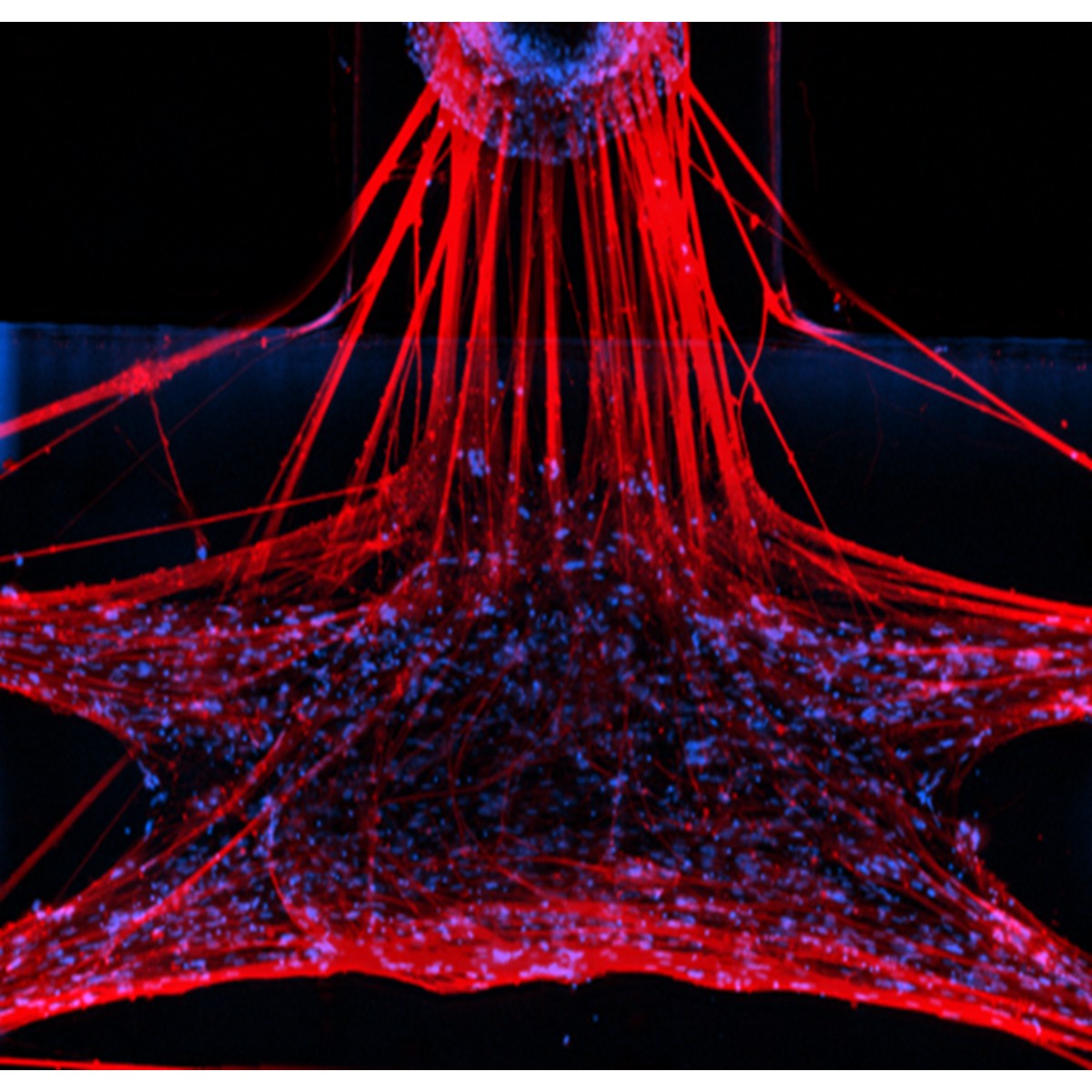
Structural and Functional Immunofluorescence Characterization of NMJ tissues
Key Capabilities
2
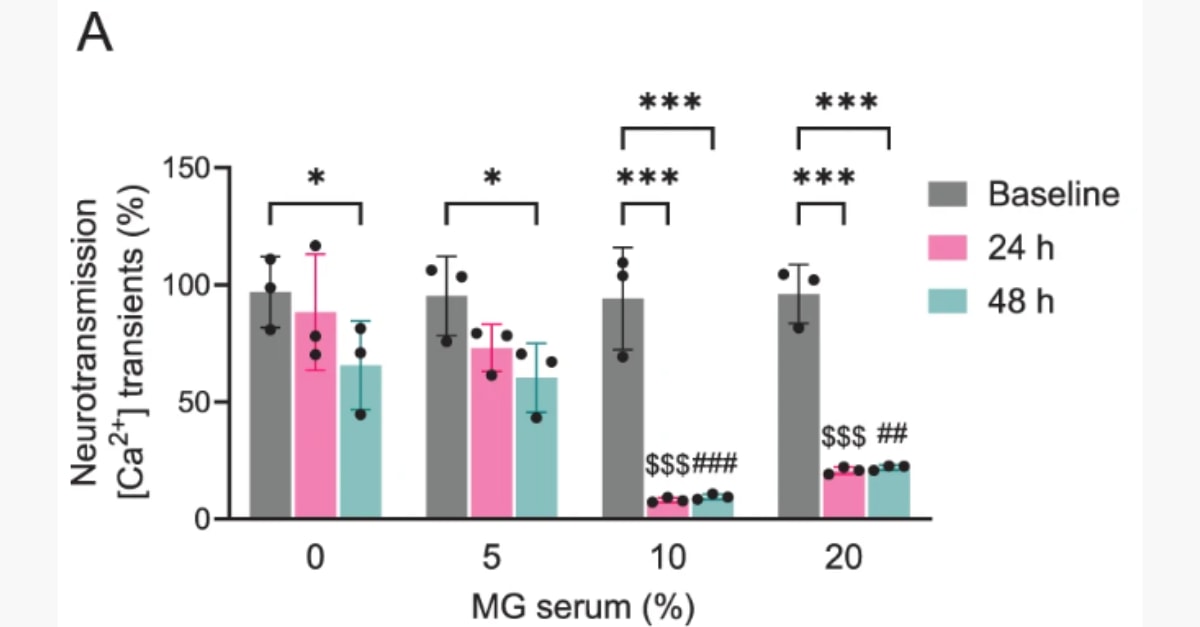
4× more sensitive and 24h faster than the mouse assay, detecting as little as 6.75 pg (0.25 Units) of Botulinum toxin A within just 24 hours.
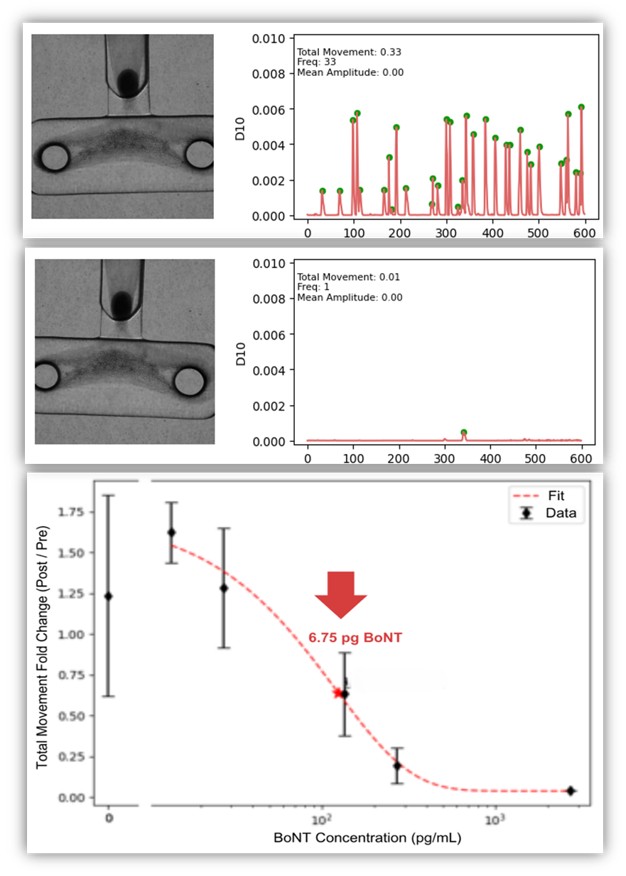
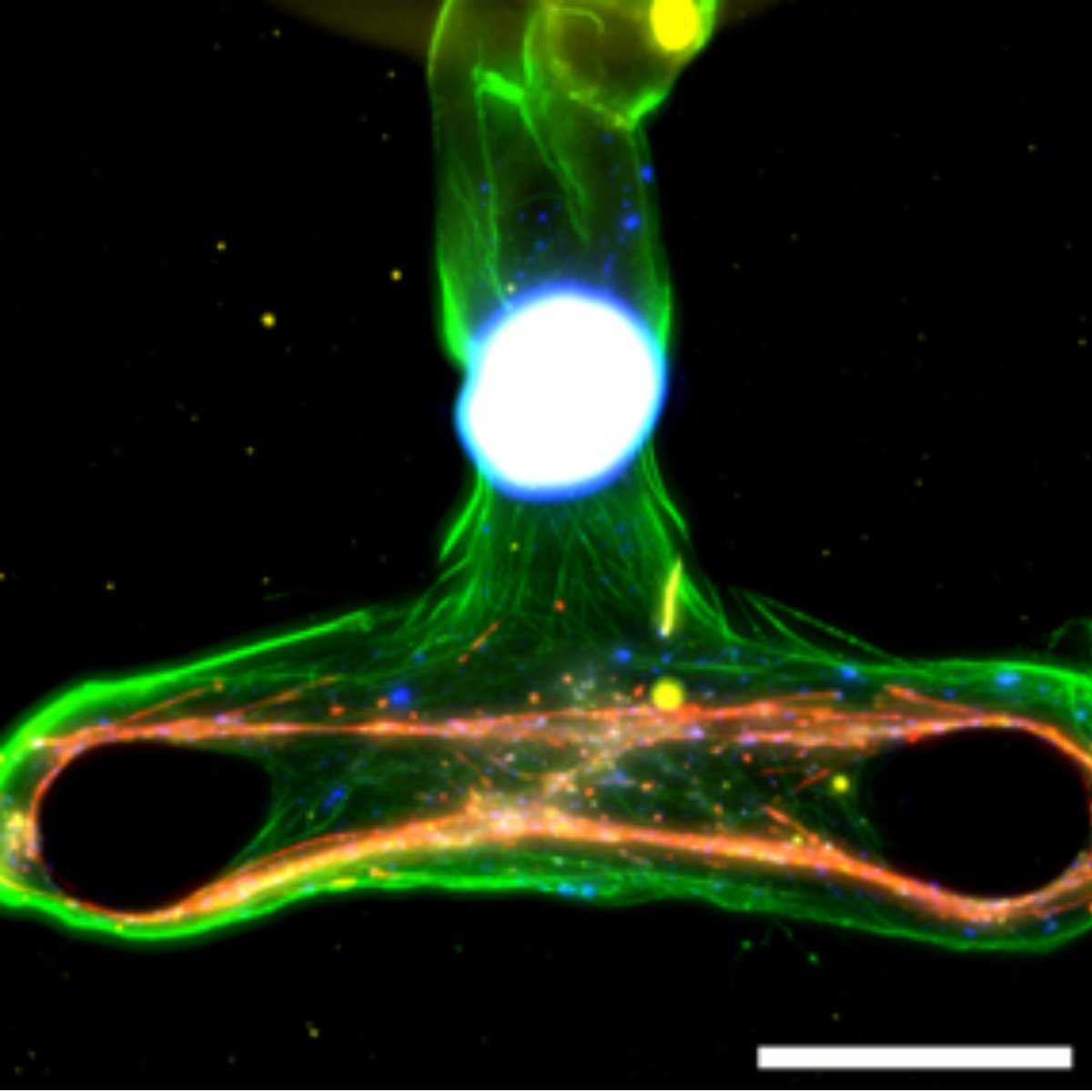
Key Capabilities
3
Enables label-free, long-term monitoring of NMJ activity (4 weeks) and live assessment of toxins, antidotes and drugs
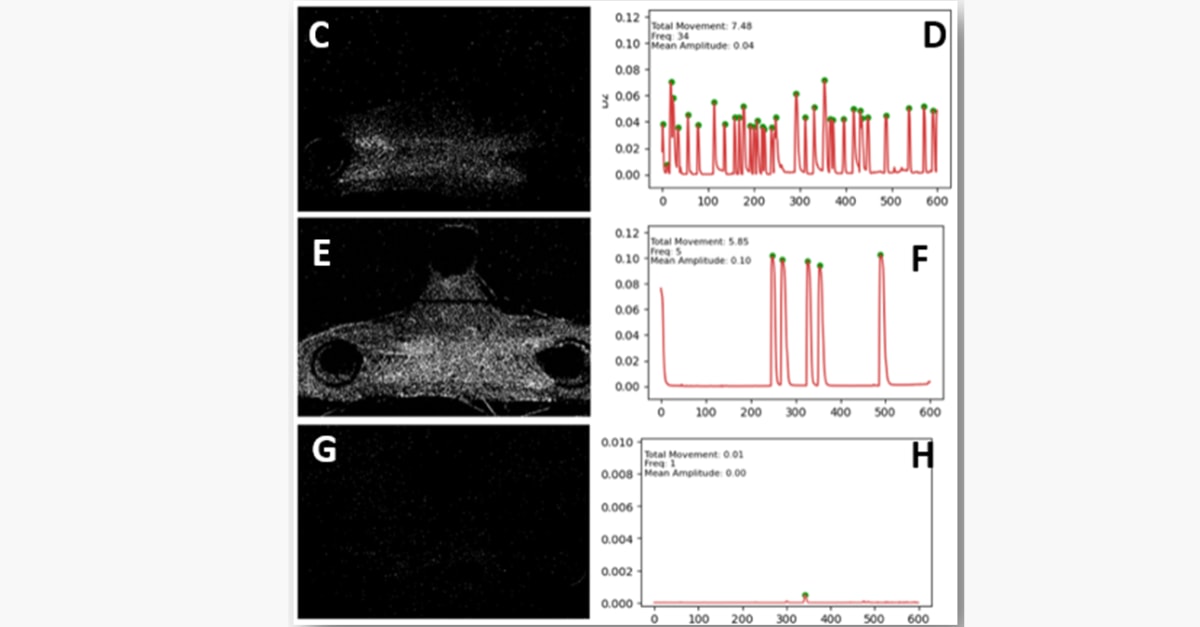
Key Capabilities
4
Enables detection of toxins and antibodies in patient-derived fluids.
Applications
NeuroMuscle™ supports a broad range of neuromuscular research, safety, and translational applications, enabling functional, human-relevant evaluation that bridges discovery, regulatory science, and human impact impact.
Neuromuscular Disease Modeling
Model neuromuscular disorders including Myasthenia Gravis, ALS, and genetic NMJ diseases using patient-derived iPSC systems, enabling analysis of NMJ damage and protective mechanisms.
Neuromuscular Toxicology & Safety
Detect and quantify neuromuscular toxicity, including botulinum neurotoxin activity, environmental neurotoxins, and pharmaceutical safety profiles using functional contractility-based assays.
Drug Discovery & Translational Research
Evaluate small molecules, biologics, and antibodies through functional efficacy, mechanism-of-action, and longitudinal therapeutic response studies in fully human NMJ systems.
Regulatory & Countermeasure Development
1
Support for regulatory-aligned testing strategies
2
Human-relevant alternatives to animal-based neuromuscular assays
3
Public health preparedness and countermeasure evaluation
4
Platform deployment in regulatory and external laboratories
Representative Use Cases
Examples demonstrating the capabilities of the NeuroMuscle™ platform. Detailed datasets and study designs are available upon request.
Request Our Sample Data
Autoimmune Neuromuscular Disease (Myasthenia Gravis)
NeuroMuscle™ has been used to model autoimmune-mediated neuromuscular junction (NMJ) dysfunction using human motor neurons and skeletal muscle. By exposing human NMJs to disease-relevant conditions, the platform captures functional loss of neuromuscular transmission and enables longitudinal assessment of therapeutic protection and recovery.
What this demonstrates
- Human-relevant modeling of NMJ pathology
- Functional readouts of neuromuscular inhibition and rescue
- Longitudinal monitoring of treatment response
Botulinum Neurotoxin (BoNT) Detection & Safety
NeuroMuscle™ enables sensitive, functional assessment of neuromuscular inhibition induced by botulinum neurotoxins in a fully human in vitro system. Changes in muscle contraction dynamics provide a direct, quantitative measure of toxin activity and recovery following intervention.
What this demonstrates
- Functional detection of neuromuscular inhibition
- Time-resolved monitoring of toxin effects
- Applicability to safety testing and countermeasure evaluation
Therapeutic Efficacy & Reversibility Studies
The platform supports longitudinal functional assessment of neuromuscular communication, allowing researchers to track onset, progression, and reversibility of drug or toxin effects over time.
What this demonstrates
- Dynamic monitoring rather than static endpoints
- Evaluation of sustained vs. transient effects
- Functional relevance in human NMJs
Regulatory & Translational Research Applications
NeuroMuscle™ has been implemented in external laboratory environments, supporting translational and regulatory-aligned research focused on human-relevant alternatives to animal-based neuromuscular assays.
What this demonstrates
- Platform robustness and reproducibility
- Ease of transfer and implementation
- Alignment with New Approach Methodologies (NAMs)

Accelerate breakthroughs with human-based organ-on-a chip solutions that deliver faster, more predictive results.
Main Page
Home
About Us
Our Platforms
Our Services
Our News
Contact Us
Our Resources
Product literature
Publications
News&Events
Webinars
Copyright© 2026 Ananda Device. inc
Terms and Conditions
Languages: EN

NeuroMuscle™
Ready-to-use. Human-based. Purpose-built for reliable neuromuscular modeling.
Signaling, Contraction &
Response — Measured in Human Models.
NeuroMuscle™ is a fully human neuromuscular junction (NMJ)-on-a-chip platform that combines iPSC-derived motor neurons and 3D skeletal muscle within a microfluidic system to recreate functional neuromuscular signaling in vitro. Designed as a plug-and-play, plate-based system, NeuroMuscle™ operates like a standard multi-well culture plate—requiring no pumps, tubing, controllers, or specialized hardware—and integrates seamlessly into existing laboratory workflows.
Each disposable plate contains 20 independent, functional NMJs, enabling parallel testing across multiple conditions while supporting high-content imaging, functional assays, and automated analysis. The platform delivers quantitative, real-time readouts of neuromuscular communication and muscle contraction, providing insights that are difficult or impossible to obtain from traditional in vitro or animal models.
NeuroMuscle™ has been successfully transferred and established in external laboratories, including regulatory and industry environments, demonstrating robustness, reproducibility, and ease of implementation. The platform supports both in-house use and CRO-led study execution, with options for ready-to-use, pre-assembled NMJs.

Key Capabilities
1
Structural and Functional Immunofluorescence Characterization of NMJ tissues

Key Capabilities
2
4× more sensitive and 24h faster than the mouse assay, detecting as little as 6.75 pg (0.25 Units) of Botulinum toxin A within just 24 hours.

Key Capabilities
3
Enables label-free, long-term monitoring of NMJ activity (4 weeks) and live assessment of toxins, antidotes and drugs

Key Capabilities
4
Enables detection of toxins and antibodies in patient-derived fluids.
Applications
NeuroMuscle™ supports a broad range of neuromuscular research, safety, and translational applications, enabling functional, human-relevant evaluation that bridges discovery, regulatory science, and human impact impact.
Neuromuscular Disease Modeling
Model neuromuscular disorders including Myasthenia Gravis, ALS, and genetic NMJ diseases using patient-derived iPSC systems, enabling analysis of NMJ damage and protective mechanisms.
Drug Discovery & Translational Research
Evaluate small molecules, biologics, and antibodies through functional efficacy, mechanism-of-action, and longitudinal therapeutic response studies in fully human NMJ systems.
Neuromuscular Toxicology & Safety
Detect and quantify neuromuscular toxicity, including botulinum neurotoxin activity, environmental neurotoxins, and pharmaceutical safety profiles using functional contractility-based assays.
Regulatory & Countermeasure Development
1
Support for regulatory-aligned testing strategies
2
Human-relevant alternatives to animal-based neuromuscular assays
3
Public health preparedness and countermeasure evaluation
4
Platform deployment in regulatory and external laboratories
Representative Use Cases
Examples demonstrating the capabilities of the NeuroMuscle™ platform. Detailed datasets and study designs are available upon request.
Request Our Sample Data
Autoimmune Neuromuscular Disease (Myasthenia Gravis)
Botulinum Neurotoxin (BoNT) Detection & Safety
Therapeutic Efficacy & Reversibility Studies
Regulatory & Translational Research Applications

Accelerate breakthroughs with human-based organ-on-a chip solutions that deliver faster, more predictive results.
Main Page
Home
About Us
Our Platforms
Our Services
Our News
Contact Us
Our Resources
Product literature
Publications
News&Events
Webinars
Copyright© 2026 Ananda Device. inc
|
Terms and Conditions
|
Languages: EN


Home
About Us
Our Platforms
Our Services
Our Resources
Contact Us
Client Portal
EN
NeuroMuscle™
Ready-to-use. Human-based. Purpose-built for reliable neuromuscular modeling.
Signaling, Contraction &
Response — Measured in Human Models.
NeuroMuscle™ is a fully human neuromuscular junction (NMJ)-on-a-chip platform that combines iPSC-derived motor neurons and 3D skeletal muscle within a microfluidic system to recreate functional neuromuscular signaling in vitro. Designed as a plug-and-play, plate-based system, NeuroMuscle™ operates like a standard multi-well culture plate—requiring no pumps, tubing, controllers, or specialized hardware—and integrates seamlessly into existing laboratory workflows.
Each disposable plate contains 20 independent, functional NMJs, enabling parallel testing across multiple conditions while supporting high-content imaging, functional assays, and automated analysis. The platform delivers quantitative, real-time readouts of neuromuscular communication and muscle contraction, providing insights that are difficult or impossible to obtain from traditional in vitro or animal models.
NeuroMuscle™ has been successfully transferred and established in external laboratories, including regulatory and industry environments, demonstrating robustness, reproducibility, and ease of implementation. The platform supports both in-house use and CRO-led study execution, with options for ready-to-use, pre-assembled NMJs.

Key Capabilities
1
Structural and Functional Immunofluorescence Characterization of NMJ tissues

Key Capabilities
2
4× more sensitive and 24h faster than the mouse assay, detecting as little as 6.75 pg (0.25 Units) of Botulinum toxin A within just 24 hours.

Key Capabilities
3
Enables label-free, long-term monitoring of NMJ activity (4 weeks) and live assessment of toxins, antidotes and drugs

Key Capabilities
4
Enables detection of toxins and antibodies in patient-derived fluids.
Applications
NeuroMuscle™ supports a broad range of neuromuscular research, safety, and translational applications, enabling functional, human-relevant evaluation that bridges discovery, regulatory science, and human impact impact.
Neuromuscular Disease Modeling
Model neuromuscular disorders including Myasthenia Gravis, ALS, and genetic NMJ diseases using patient-derived iPSC systems, enabling analysis of NMJ damage and protective mechanisms.
Drug Discovery & Translational Research
Evaluate small molecules, biologics, and antibodies through functional efficacy, mechanism-of-action, and longitudinal therapeutic response studies in fully human NMJ systems.
Neuromuscular Toxicology & Safety
Detect and quantify neuromuscular toxicity, including botulinum neurotoxin activity, environmental neurotoxins, and pharmaceutical safety profiles using functional contractility-based assays.
Regulatory & Countermeasure Development
1
Support for regulatory-aligned testing strategies
2
Human-relevant alternatives to animal-based neuromuscular assays
3
Public health preparedness and countermeasure evaluation
4
Platform deployment in regulatory and external laboratories
Representative Use Cases
Examples demonstrating the capabilities of the NeuroMuscle™ platform. Detailed datasets and study designs are available upon request.
Request Our Sample Data
Autoimmune Neuromuscular Disease (Myasthenia Gravis)
Botulinum Neurotoxin (BoNT) Detection & Safety
Therapeutic Efficacy & Reversibility Studies
Regulatory & Translational Research Applications

Accelerate breakthroughs with human-based organ-on-a chip solutions that deliver faster, more predictive results.
Main Page
Home
About Us
Our Platforms
Our Services
Our News
Contact Us
Our Resources
Product literature
Publications
News&Events
Webinars
Copyright© 2026 Ananda Device. inc
|
Terms and Conditions
|
Languages: EN

Home
About Us
Our Platforms
Our Services
Our Resources
Contact Us
Client Portal
EN